CellCreep Biotech is a biotech company promoted by a group of scientists became entrepreneur with a Vision of providing quality human biologics, especially biosimilars. We have international partners to procure Biosimilars, Probiotics & Biopesticides products and technologies and make the biotech firms to form a full function or biologic production network.
We have expertise in product development using various platforms including E. coli, Yeast & CHO based wide range of biotech products including Growth factors, Hormones and Monoclonal antibodies (mAb’s) and fusion proteins. CellCreep Biotech also Procure Probiotics, Biopesticides products/technologies.
CellCreep Biotech has international partners (Asia pacific & Indian CRO Companies) to procure Biosimilar products and technologies and make the biotech firms to form a full function or biologic production network. The main goal is to help translating the research finding to biologic products meeting the world market and regulatory needs.
CellCreep Biotech has well-trained technical team who are specialized in biopharmaceutical process development and manufacturing to help & accelerate the preclinical & clinical trial batches in budget saving and timely fashion and minimize expenses for the clients.
CellCreep Biotech is currently collaborated with following reputed Biotech CRO & Incubation centres & Universities
VIT University (School of Bioscience and Technology – SBST),Vellore, India.
Reputed CRO Companies in Asia-Pafic Countries, India, Bangalore.
Kristhu Jayanthi College (Autonomous), Bangalore, India.
CellCreep Biotech will procure complete package of Technology Transfer (Process training in client's site), Process development, Analytical method development with final product certification, Scale-up, Pre-clinical, Clnical trial & cGMP Manufacturing supports as a Turnkey project.
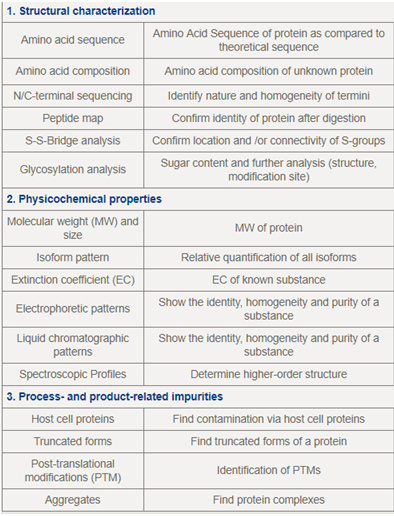
CellCreep Biotech has strategic partnership with a leading Biosimilars Proteomics Contract Analytical Services company in UK & Germany, who will procurethe analysis results as per the program in accordance with ICH Q6B guidelines (Including method setup, validation of customer-specific methods and release testing) to qualify multiple regulatory bodies worldwide including USFDA, EMA during all stages of biosimilars development: Scientific advice, phase I, phase III to Marketing Authorization.
Biosimilars - Protein Analytics & Certification(Biosimilarity/Comparability)
We will procurethe analysis results as mentioned in the table, as per the program in accordance with ICH Q6B guidelines
CellCreep Biotech has international partners to procure Biosimilars, Probiotics& Biopesticides products and technologies and make the biotech firms to form a full function or biologic production network.